Culture Conditions Mono Mac 1 and Mono Mac 6
Cell culture
Starting from a frozen sample, the 1 ml cell suspension is diluted to 10 ml with culture medium right after thawing, cells are spun once, resuspended in culture medium and are seeded in 24 well plates at about 5 x10 5 /ml with 2ml per well and cultured overnight. Following this initial step, cells are seeded at a density of 2 x 10 5 /ml in 2 ml volumes in 24-well-plates and they are cultured at 37°C in a humidified 5% CO 2 atmosphere for 3-4 days before being used for experiments or before being pooled, counted and reseeded again (for washing spin at 400 g for 7 min). Doubling time is approx. 2 days. The culture medium is RPMI 1640 with 10-20 % fetal calf serum, fortified with several additives as detailed below.
The cultures have to be monitored on a regular basis for mycoplasma contamination and for the expression of the CD14 antigen (e.g. My4 antibody).
Upon receipt of the cell line, it is recommended to first establish a batch of frozen aliquots, which can be used to retrieve the original cells at regular time intervals or in case problems like contamination arise. For freezing of viable cells use 4-10 x 10 6 cells per ml in RPMI 20 % FCS and add dropwise an equal volume of RPMI 20 % FCS, 20 % dimethyl sulfoxide (DMSO), while swirling the tube in ice water. For optimal freezing, a controlled freezer device is recommended. Alternately, freezing may be tried by putting the tubes in a well insulated styrofoam box into a -80° C freezer overnight. Then transfer to liquid nitrogen (or storage at least at -135° C).
Composition of culture medium for Mono Mac 6
RPMI 1640 L-Glutamine or L-Alanine-LGlutamine, use at 2 mM final
Penicillin Streptomycin 5000 U/mL 100X
Non-Essential Amino Acids (NEAA) 100X use at 1-2 X final
OPI-supplement 10 ml for 1 ltr (Sigma-Aldrich # O - 5003) contains oxalacetic acid, sodium pyruvate and insulin
Optional: Filtration to remove LPS (see below), then add fetal calf serum (FCS) at 10-20% final.
The FCS needs to be LPS free, batches should be tested for best culture performance before purchase.
Removal of LPS
In case of contamination of culture medium with lipopolysaccharide, the Mono Mac 6 cells will grow slower and will clump. A typical pattern when inspecting the 24 well plate under the inverted microscope looks as follows:
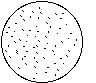
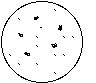
no LPS LPS (e.g. 10 nanog/ml)
In order to avoid introduction of LPS, use quality reagents only.
If required, LPS may be removed by ultrafiltration using a 20 kD cut off column.
Here, ULTRA SteriSet #103564, available at Baxter Renal Care, can be employed.
For this procedure, fetal calf serum (with many proteins at > 20 kD) cannot be included in the filtration process. It has to be added after filtration and therefore needs to be free of LPS (or other pyrogens) to begin with. Absence of LPS in a batch of culture medium may be verified by the Limulus assay or by the cell clumping in 24 well plates (see Figure).
Establishing a LPS-resistant cell line
In case you are not interested in LPS induced gene expression, you may generate a LPS-resistent line by feeding the cells with gradually increasing doses of LPS (Salmonella minnesota, L-6261, Sigma) from 0.5 ng/ml to 1 ug /ml, which can be done within 3 weeks time. You then always supplement your culture medium with 1 ug LPS / ml.

