Markers and Staining Protocols Human Monocytes
Introduction
Since its initial description in 1989 (Passlick et al, Blood, 1989) human monocyte subsets have gained increasing attention and their behavior and role in health and disease has been described in more than 1.000 publications (see CD14+CD16+ literature section of this web-page). Still, many studies lack a robust approach for exact determination of these monocytes. Recently an international panel under the auspices of the IUIS and the WHO has presented a nomenclature proposal for monocytes and dendritic cells, including human monocytes (Ziegler-Heitbrock et al, Blood 2010). The proposals subdivides the human monocytes into classical CD14++CD16- monocytes and non-classical CD14+CD16++ monocytes. In addition intermediate CD14++CD16+ monocytes are defined. Described below is a typical flow cytometry approach that combines a 4 color analysis with absolute counting beads for definition and enumeration of the classical and non-classical monocytes and also intermediate monocytes (Method A, Heimbeck et al, Cytometry, 2010). Furthermore, for better definition of the intermediate monocytes we have described an approach that uses the slan-marker for separation of slan-negative intermediate monocytes and slan-positive non-classical monocytes (Method B, Hofer et al, Blood, 2015)
METHOD A Reagents
CD45 Allophycocyanin (APC)-conjugated (# IM2473, isotype: mouse IgG1)
Anti-HLA-DR PC5-conjugated (# PM 2659U, isotype: mouse IgG1)
CD14 fluorescein isothiocyanate (FITC)-conjugated clone My4 (# 6603511, isotype: mouse IgG2b)
CD16 phycoerythrin (PE)-conjugated clone 3G8 (# A07766, isotype: mouse IgG1). All antibodies and their respective
isotype controls are from Beckman Coulter, Krefeld, Germany.
Absolute counting beads: CountBright absolute counting beads® (# C36950, Invitrogen, Karlsruhe, Germany).
METHOD A Blood sampling and staining procedure
Blood is collected in K3E S-Monovettes® (# 05.1167 Sarstedt, Germany) and 100 µl of the whole blood is admixed with 5µl of each antigen specific fluorochrome–labelled antibody (dilution 1:20). The samples are then incubated for 20 min at 4°C in the dark. Lysis of erythrocytes is performed using a Coulter Q-Prep® lysis instrument, which adds a volume of 1000 µl, and the sample is supplemented with 100µl of CountBright absolute counting beads for determination of absolute numbers of cells. To ensure maximum viability, stained cells are analyzed promptly.
METHOD A Flow cytometry gating strategy
All signals are acquired on a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA). The instrument amplifier setting was calibrated monthly using Calibrite™3 beads and Calibrite™APC beads (# 340486 and # 340487, BD Biosciences). CellQuest software (version 3.2, 1998) is used to acquire data. First, the CD45-positive leucocytes are visualized in a histogram showing all fluorescence 4 (APC-positive) events. An acquisition threshold is set such that any unwanted events like CD45-negative platelets, dead cells and debris are not recorded.
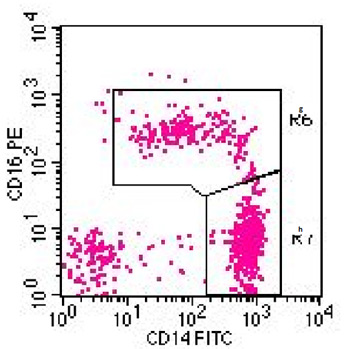
The monocytes are then defined by sequential gating on all CD45-positive leukocytes in light scatter plots, CD14 versus HLA-DR-staining and CD14 versus CD16 staining. Using isotype controls, voltage and compensation the instrument is set such that the cells are adequately positioned in the dot plots.
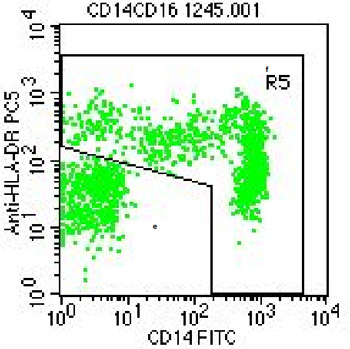
For direct determination of absolute numbers of cells, we use CountBright® absolute counting beads (Invitrogen, Karlsruhe, Germany). These beads are visualized in the CD45 plot above as a narrow peak at the upper end of the scale.
At least 1000 CD14+CD16++ events are acquired per sample. The number of absolute CD14+CD16++ monocytes are then calculated with reference to the flow count beads (50 ul added) according to the following formula (for details refer to bead provider user instructions):
Using an isotype control for CD16 the can be used to clearly dissect the CD14++CD16+ monocytes from the CD14++CD16- classical monocytes (see example below). These CD14+CD16++ intermediate monocytes in healthy individuals form a very minor population of cells, but they can become a major population in inflammatory diseases like asthma and sarcoidosis.
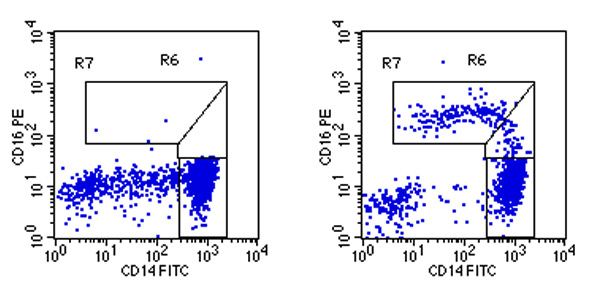
For further details and clinical results with this assay please see:Heimbeck, I, Hofer, TPJ, Eder, C, Adam Wright, A, Frankenberger, M, Marai, A, Boghdadi, G, Scherberich, J, Ziegler-Heitbrock, L: Standardized single-platform determination of human monocyte subpopulations, Cytometry, 77A: 823, 2010
METHOD B Reagents
SLAN-FITC IgM, # 130-093-027 Miltenyi-Biotec; Isotype-FITC IgM, #130-093-178 Miltenyi-Biotec; CD16-PE IgG1 clone 3G8, # A07766, Beckman Coulter; , CD14-PC5 IgG2b clone My4, # A07765 Beckman Coulter, HLA-DR-APC IgG1, # IM3635, Beckman Coulter; all antibodies are use at 1:20 final dilution. Counting beads (C36950; Molecular Probes).
METHOD B Blood sampling and staining procedure
Blood is collected in K3E S-Monovettes® (# 05.1167 Sarstedt, Germany) and within 4 hs 100 µl of the whole blood is admixed with 5µl of each antigen specific fluorochrome–labelled antibody (dilution 1:20). The samples are then incubated for 20 min at 4°C in the dark. Lysis of erythrocytes is performed using a Coulter Q-Prep® lysis instrument, which adds a volume of 1000 µl, and the sample is supplemented with 100µl of Molecular Probes absolute counting beads for determination of absolute numbers of cells. To ensure maximum viability, stained cells are analyzed promptly.
METHOD B Flow cytometry gating strategy
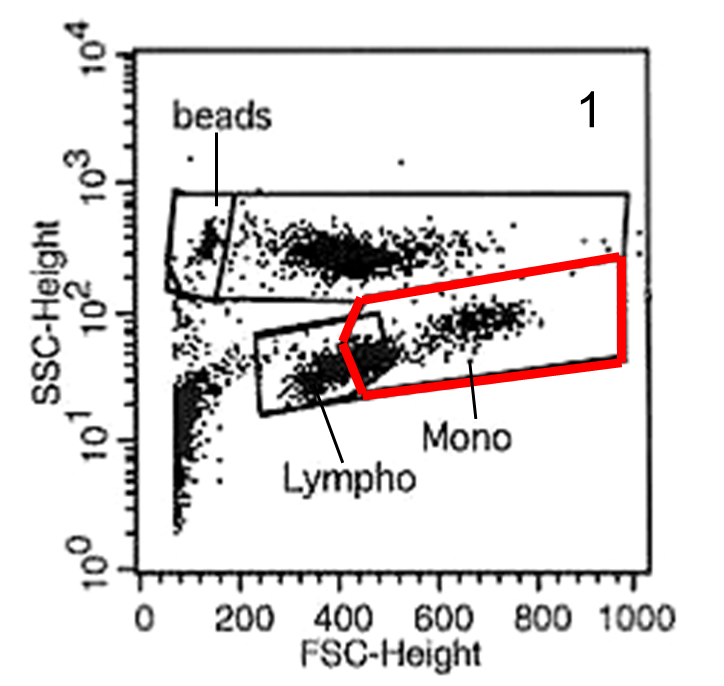
All signals are acquired on a FACSCalibur flow cytometer (BD Biosciences, San Jose, CA). CellQuest software (version 4.0.2) is used to acquire data. First the events are defined in the forward scatter (FSC, linear scale) versus side scatter (SSC, log scale) plot. A gate is put around the absolute counting beads and around the monocytes including the upper third of the lymphocytes, since the latter include a portion of the non-classical monocytes (see Figure1)
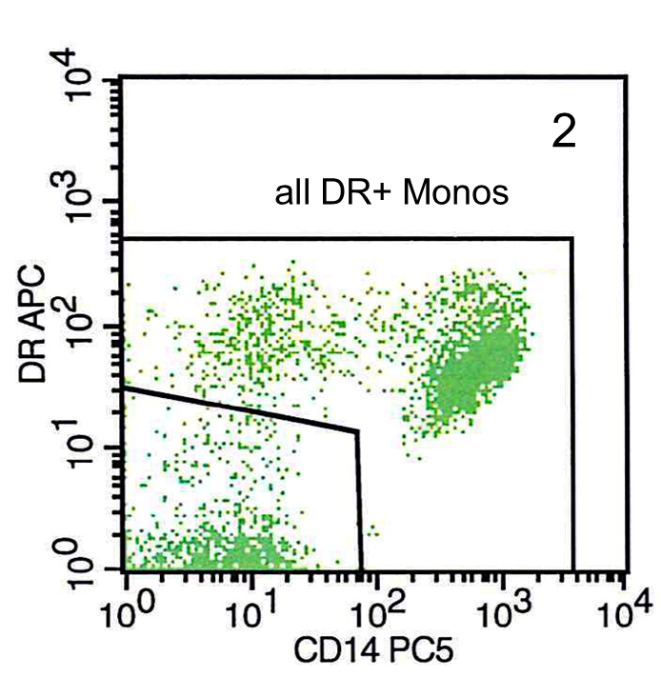
The cells from the monocyte scatter gate are then shown in the CD14versus HLA-DR plot. Here a gate is put around all CD14+DR+ events giving all DR+ cells, which still includes DR+CD14- cells, like B cells and activated T cells (Figure 2).
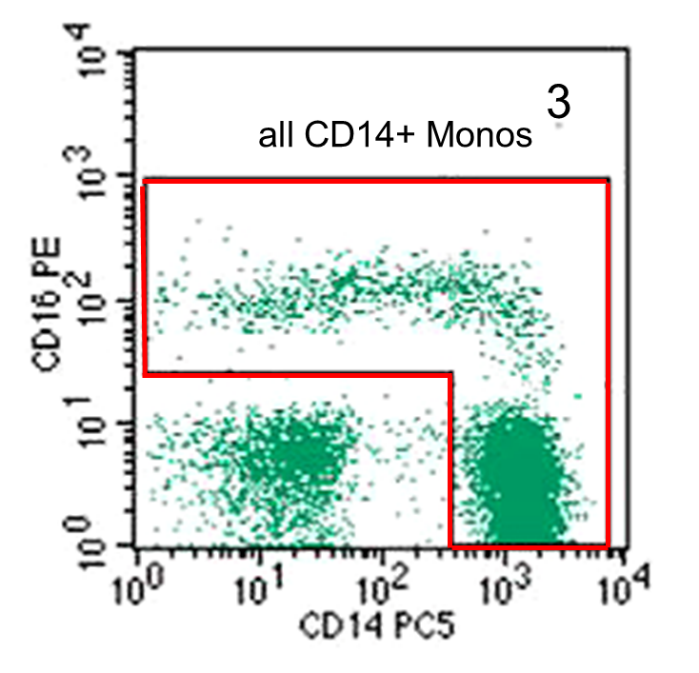
These events are then shown in the CD14 versus CD16 plot, which is used to determine all monocytes as shown in the red gate in Figure 3
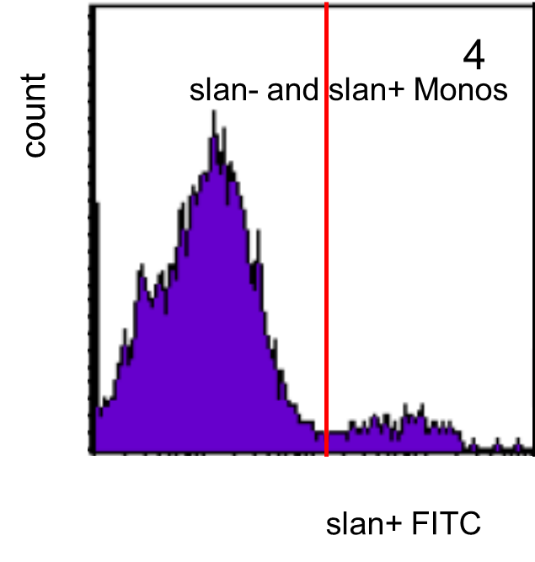
For these cells the slan staining is shown in a single color histogram and here the slan+ cells are defined by a cut-off line as shown in Figure 4.
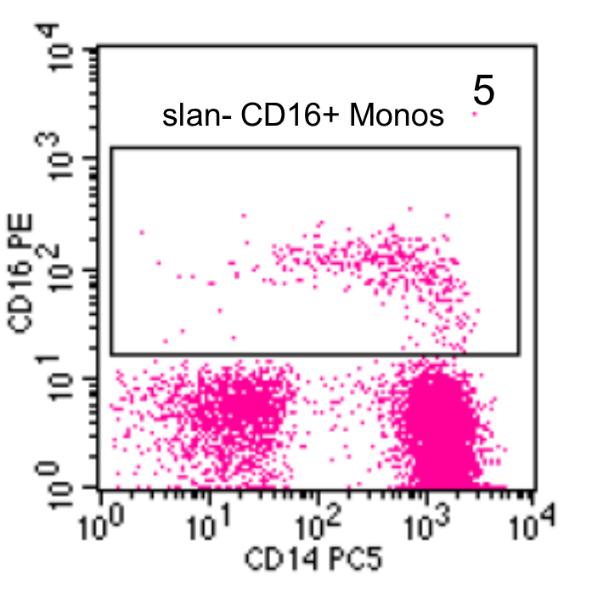
The slan-negative cells are then shown in a CD14 versus CD16 dot plot and a gate is set around the CD16+ cells (Figure 5). These are the slan-negative CD16+ intermediate monocytes.
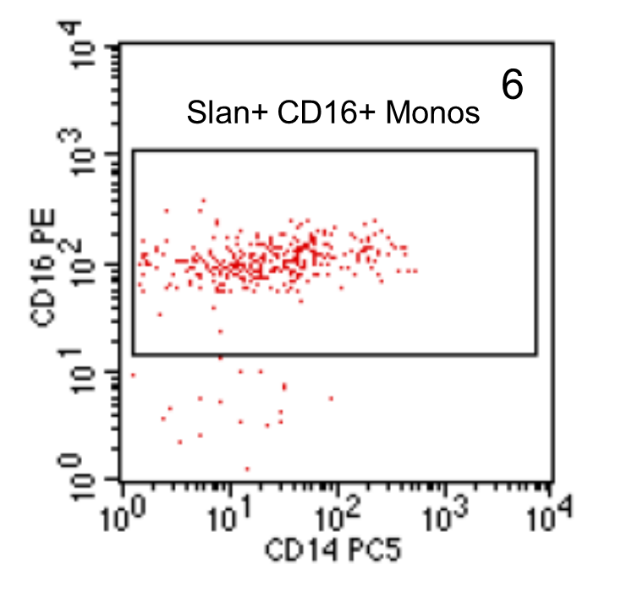
Then the slan-positive cells are shown in a CD14 versus CD16 dot plot and a gate is set around the CD16+ cells (Figure 6). These are the slan+ CD16+ non-classical monocytes.
For direct determination of absolute numbers of cells, we use absolute counting beads (Molecular probes). These beads are visualized in a FITC versus PE plot gated on the beads gated in the intial scatter plot in Figure 1
At least 500 slan+CD16+ events were acquired per sample. The absolute number of slan-positive non-classical monocytes is then calculated with reference to the flow count beads (50 ul added) according to the following formula (for details refer to bead provider user instructions):
An isotype control for CD16 can be used to clearly dissect the CD16+ monocytes from the CD14++CD16- classical monocytes and for the slan marker an IgM-isotype control can be used to help setting the cut-off.
For further details and clinical results with this assay please see: Hofer, TP, Zawada, AM, Frankenberger, M, Skokann, K, Anna A. Satzl, AA, Wolfgang Gesierich, W, Madeleine Schuberth, M, Johannes Levin, J, Adrian Danek , A, Rotter, B, Heine, GH, Ziegler-Heitbrock, L: slan-defined subsets of CD16-positive monocytes: Impact of granulomatous inflammation and M-CSF-receptor mutation, Blood,126:2601-2610, 2015

